Download the following resources to help you fight Big Marijuana
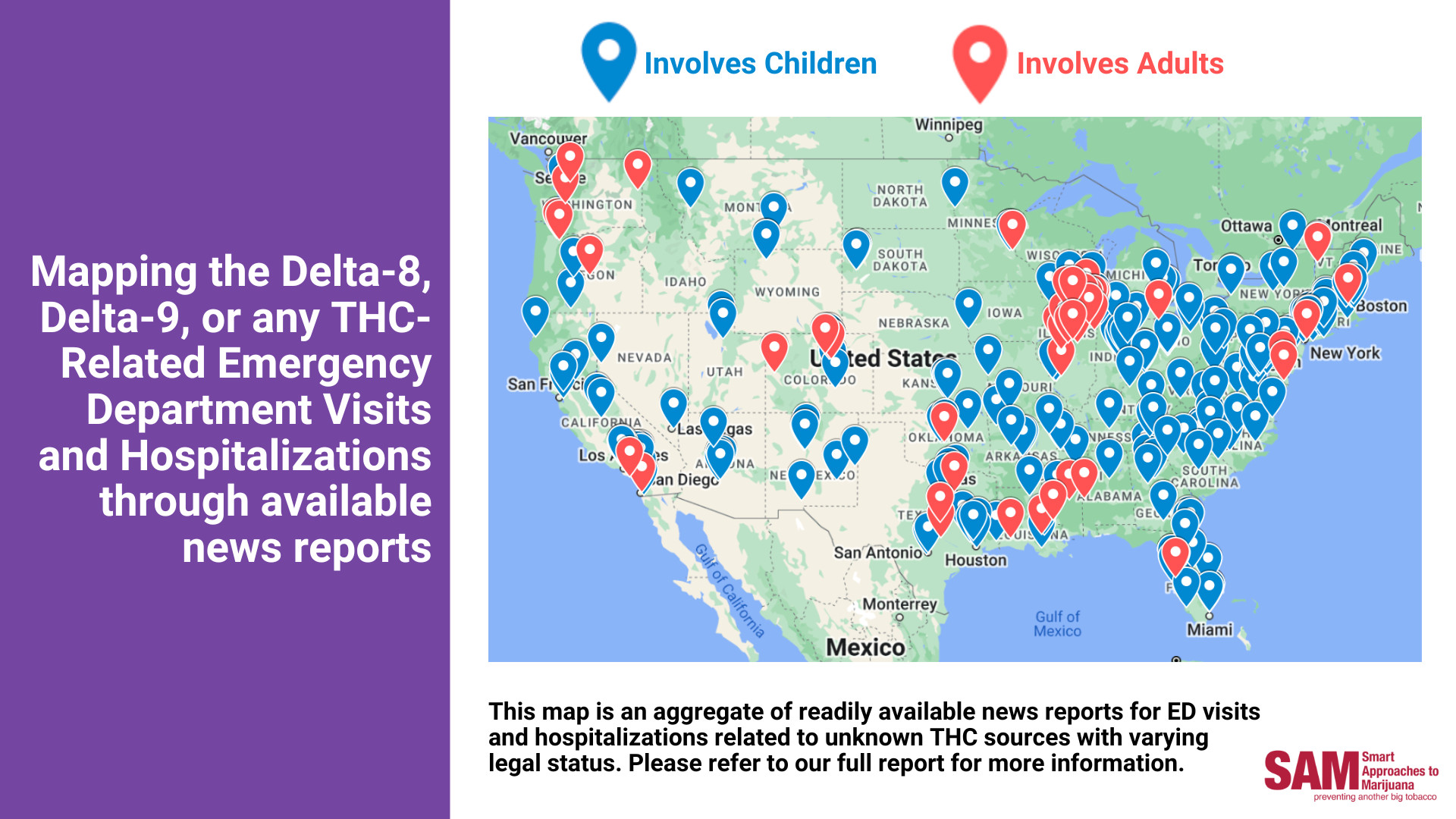
Mapping the Delta-8, Delta-9, or any THC-Related Emergency Department Visits and Hospitalizations through News Reports
Read More

Driving Under the Influence of Drugs. THC Laws are Flawed
Watch Video
The Unforeseen Consequences of Rising THC Levels in Marijuana: A Growing Public Health Concern
Download Toolkit

Learn more about high potency marijuana & the need for THC potency caps.
Download Toolkit
Legalizing and taxing marijuana won’t solve COVID-19 budget issues.
Download Toolkit

Are states with legal marijuana bringing in massive tax revenues? No.
Download Toolkit

Has marijuana legalization benefited the movement for social justice? No
Download Toolkit



